![]()
Biopharmaceutical Sciences, Biomed Biopharm Res., 2021; 18(2):228-237
doi: 10.19277/bbr.18.2.271; [+] PDF version here; [+] Portuguese html version here
Rapid UV-Vis spectroscopy methods for quantification of ranitidine tablets
Patrícia Rijo 1,2*, Lara Ribeiro 1, Marisa Nicolai 1, Paula Pereira 1,3,4, Célia Faustino2*
1 Center for Research in Biosciences & Health Technologies (CBIOS), Universidade Lusófona de Humanidades e Tecnologias, 1749-024 Lisboa, Portugal;
2 Research Institute forMedicines (iMed.ULisboa), Faculty of Pharmacy, Universidade de Lisboa, Av. Prof. Gama Pinto, 1649–003, Lisbon, Portugal;
3 Center for Natural Resources and Environment (CERENA), Instituto Superior Técnico (IST), Universidade de Lisboa, Av. Rovisco Pais, 1049-001 Lisboa, Portugal;
4 EPCV-ULHT-Universidade Lusófona de Humanidades e Tecnologias, 1749-024 Lisboa, Portugal
*corresponding authors:
Abstract
Ranitidine is a histamine H2-receptor antagonist that inhibits gastric acid secretion, commonly used in the treatment and prophylaxis of peptic ulcer and gastroesophageal reflux disease. Over-the-counter ranitidine hydrochloride tablets are available in several European countries as well as the US for the relief of indigestion and heartburn. Commercial tablets containing the equivalent of 150 mg of ranitidine were analysed following extraction. Ranitidine is water-soluble, thus avoiding the use of organic solvents. Standard aqueous solutions of ranitidine hydrochloride were used to construct a calibration curve. This graduate laboratory experiment describes a simple, inexpensive, safe, and “green” procedure for the quantification of ranitidine tablets by UV spectrophotometry. The entire procedure provides an pedagogical opportunity to approach a realistic analytical scenario, typical of a quality control laboratory, by designing an attractive and engaging hands-on session. An average content of 148 ± 1 mg of ranitidine was determined using both standard addition and external calibration methods and results are in agreement with Pharmacopoeia.
Keywords: Analytical chemistry, ranitidine, drugs/pharmaceuticals, quantitative analysis, UV-Vis spectroscopy
Received 01/09/2021; Accepted: 21/12/2021
Introduction
UV-Vis spectroscopy is a feasible and straightforward analytical tool routinely used with other assay techniques in the pharmaceutical industry in areas such as quality control and quality assurance (1). Several papers describing the quantification of analytical samples by UV spectrophotometry in the pharmaceutical, biotechnological, and environmental areas, as well as in the field of food and beverage, have been reported in the literature (2-21).
Highly conjugated organic compounds, including many drugs and biological macromolecules (e.g., proteins and nucleic acids) absorb light in the ultraviolet (UV)-visible (vis) region of the electromagnetic spectrum. Quantitative analysis is based on the relationship between the degree of absorption and the concentration of the absorbing species in solution, mathematically described by the Beer–Lambert law, A = elc, where A is the absorbance at a fixed wavelength, e is the absorption coefficient, l is the path length and c is concentration.
This experiment was conceived to introduce the technique of ultraviolet spectroscopy to undergraduate students in Pharmaceutical Organic Chemistry classes and describes a simple, inexpensive, and environmentally green (organic solvent-free) laboratory procedure for the quantitative assay of ranitidine, a histamine H2-receptor antagonist, in commercial 150 mg ranitidine tablets.
Ranitidine is a histamine H2-receptor antagonist that inhibits gastric acid secretion induced by histamine, pentagastrin, and other secretagogues. Ranitidine hydrochloride (Figure 1) is a white or pale yellow, water-soluble, crystalline powder.

Ranitidine is commonly used in the treatment and prophylaxis of peptic ulcer and gastroesophageal reflux disease, and also in other conditions where reduction of gastric secretion and acid output is desirable. It can also be administered with nonsteroidal anti-inflammatory drugs (NSAIDs) to reduce the risk of ulceration. The usual dose of ranitidine is either 150 mg twice a day or 300 mg once a day, usually at night.
Several preparations of ranitidine hydrochloride are available over the counter (OTC) for the relief of indigestion and heartburn, including 150 mg tablets. Each tablet for oral administration contains 168 mg of ranitidine hydrochloride (equivalent to 150 mg of ranitidine anhydrous free base).
Students are provided with 150 mg ranitidine tablets and a stock solution of ranitidine hydrochloride for the preparation of ranitidine standard solutions to be used in the construction of a calibration curve. The ranitidine tablets must be subjected to pre-analytical treatment for selective extraction and dilution of ranitidine for subsequent quantification.
The mass (mg) of ranitidine in the prescription tablet is determined from the calibration curve obtained, and the value is then compared with the label claim of the manufacturer according to the specifications of the Pharmacopeia. The official monographs of the European Pharmacopoeia (22) (and also the USP (23)) require that ranitidine tablets contain an amount of ranitidine hydrochloride (C13H23ClN4O3S) equivalent to not less than 90.0 % and not more than 110.0 % of the (package) labelled quantity of ranitidine (C13H22N4O3S) (24).
The entire laboratory procedure is completed in a three-hour lab session and is ideally performed by students working in group. It provides students with an opportunity to solve an authentic analytical problem with a commercial pharmaceutical sample in a realistic and engaging lab session. Although originally intended for undergraduate Pharmaceutical Organic Chemistry students, the experiment is simple enough to be performed by undergraduate students within a general chemistry course. It is also suited for advanced high school chemistry students, as long as a suitable UV spectrophotometer is available.
Materials and Methods
Reagents
Ranitidine hydrochloride was obtained from Sigma-Aldrich and used as received. OTC ranitidine hydrochloride tablets labelled to contain 150 mg of ranitidine were purchased from a local pharmacy. Absorbance measurements were performed ten times for blanks and in triplicate for standards and sample at 25 °C with an ultraviolet-visible spectrophotometer (Shimadzu UV-1603, Kyoto, Japan) using 1 cm length quartz cells. Distilled water (solvent) was used as the reference blank.
Experimental procedure
Standard addition and external calibration methods were performed to quantify ranitidine in commercial tablets.
Firstly, for the external calibration method, ranitidine hydrochloride was dissolved in water in order to prepare a 500.0 µg mL-1 stock solution, which was provided to the students. From the stock solution, students performed serial dilutions to prepare 25 mL ranitidine hydrochloride standard solutions with concentrations in the range 1.00–20.00 µg mL-1 to be used in the construction of a calibration curve.
The preparation of the pharmaceutical sample was based on the assay described in Pharmacopeia (22, 23) for ranitidine tablets. Five 150 mg ranitidine tablets were accurately weighed and crushed with a mortar and pestle. The resulting powder, equivalent to the average mass of one tablet, was dissolved in distilled water, with gentle stirring, to a final volume of 500 mL. The solution was filtered and a 1:100 dilution of the filtrate was performed, leading to the pharmaceutical sample to be analysed, with theoretical ranitidine concentration of 3.00 µg mL-1
For the standard addition method, 2.50 mL of the previously prepared sample solution (3.00 µg mL-1) was added to a 25 mL volumetric flask and the standard solutions were mixed and diluted to (final) ranitidine hydrochloride concentrations in the range of 1.0–20.0 µg mL-1.
The UV absorption spectra of the ranitidine hydrochloride stock solution and pharmaceutical sample were recorded between 400 and 200 nm, using distilled water as the reference blank, to compare the two profiles and determine the wavelength(s) of maximum absorbance. Absorption measurements of ranitidine hydrochloride solutions were performed in triplicate at the wavelengths of maximum absorption.
For accurate and precise quantification of the ranitidine content, the limit of detection (LOD) and the limit of quantification (LOQ) were determined (25). The LOD was determined using Equation 1, and the LOQ by Equation 2:
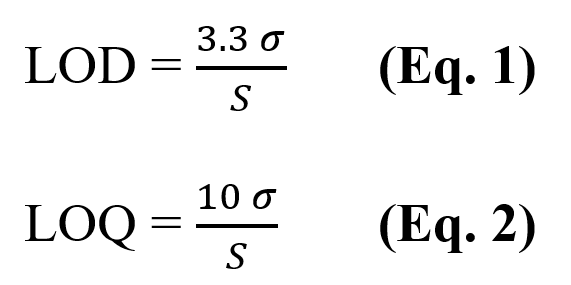
where σ is the standard deviation of the absorbance readings of the blank and S is the slope of the calibration curve.
Thus, ten absorbance measurements of the blank were performed at the wavelength of maximum absorbance of ranitidine to ensure a relative standard deviation (RSD) of less than 10%, as recommended by IUPAC (26).
Results and Discussion
The UV absorption spectra of the ranitidine hydrochloride stock solution and pharmaceutical sample solution are shown in Figure 2.
Figure 2 - UV absorption spectrum of a 10.0 µg mL−1 ranitidine hydrochloride stock solution (upper curve) and a solution from a commercial sample (lower curve) registered between 400 and 200 nm.

Sample and stock aqueous solutions show two absorption peaks at 314 nm and 227 nm. Absorption measurements are then performed at 314 nm and 227 nm against distilled water as the reference blank. In order to quantify ranitidine in tablets, students must convert the amount of ranitidine chloride to the ranitidine free base. In solution, the absorbing species is ranitidine, with hydrogen chloride present (only) to stabilize the ranitidine molecule against oxidation. Typical average absorption values and corrected ranitidine concentrations are shown in Table 1.
From data shown in Table 1, calibration curves were prepared (Figure 3) by plotting absorption values against ranitidine concentration. A linear relationship between absorbance and concentration was observed in the studied concentration range of 0.9–17.9 µg mL-1, thus following the Lambert-Beer law.

The equations of this straight line could be obtained from regression analysis using either a computer program or a scientific or graphing calculator in order to determine the ranitidine concentration (µg mL-1) in the sample solution and in the original pharmaceutical tablet, taking into account the dilution factor (Table 2).
Table 2 - Quantification range, equation of calibration curve, square of correlation coefficient, limit of detection (LOD), limit of quantification (LOQ), ranitidine mass, relative error at 227 nm and 314 nm for external calibration and standard addition methods.


The mass of ranitidine (mg) in the prescription tablet was then determined from the concentration of ranitidine (mg/tablet) in the pharmaceutical sample and the value obtained is compared with the label claim of the manufacturer and with the specifications of the European Pharmacopoeia for ranitidine tablets. Students’ average results was 148 ± 1 mg of ranitidine, equivalent to a good agreement with the tolerable and expected percentage range of the labelled claim as set forth by Pharmacopoeia (22, 23).
Conclusion
The experimental procedure presented for quantification of the water-soluble drug ranitidine, a histamine H2-receptor antagonist, represents a non-hazardous, simple, and reproducible laboratory experiment that can be conducted at an undergraduate level.
The use of UV spectrophotometry allowed for the quantification of ranitidine in the tablets analysed. An average content of 148 ± 1 mg of ranitidine was calculated, in good agreement with the amount claimed on the product label and in accordance with the specifications of the European Pharmacopoeia. The ratio, between the maximum absorbances at 227 nm and 314 nm, were 1.05 and 1.07 for standard stock and sample solutions, respectively, also in conformity to Pharmacopoeia (22,23). The methods used were found to be simple, rapid, accurate, precise, and easily implemented in a three-hour laboratory class. Moreover, the requirement of readily available reagents that are inexpensive and safe to handle makes the analytical method economical and environmentally friendly. The proposed quantitation of ranitidine from commercial tablets can be made at 227 nm and/or at 314 nm using an external standard method and/or the standard addition method, showing relative error in the same order of magnitude.
Authors Contributions Statement
P.R., L.R., M.N., P.P., C.F. - conceptualization and study design; M.N. - experimental implementation; M.N., P.P. - data analysis; M.N., P.P. - drafting, editing and reviewing; M.N- figures and graphics; P.R., L.R., M.N., P.P., C.F - supervision and final writing.
Funding
This research was funded by Fundação para a Ciência e a Tecnologia (FCT, Portugal), through projects UIDP/04567/2020 and UIDB/04567/2020. P.P. Gratefully acknowledges the support of the CERENA strategic project FCT-UID/ECI/04028/2019.
Acknowledgments
The authors acknowledge all the participants.
Conflict of interest
The authors declare that there is no financial or personal relationship that could present a potential conflict of interests.
References
- Görög, S. (1995). UV-visible spectrophotometry in pharmaceutical analysis. CRC Press, Boca Raton, 1995.
- Egts, H., Durben, D. J., Dixson, J. A., & Zehfus, M. H. (2012). A Multicomponent UV Analysis of α- and β-Acids in Hops. Journal of Chemical Education, 89,117–120.
- Kimaru, I. W., Zhao, F., & Chichester, K. (2010). Spectrophotometric quantification of ibuprofen release in dissolution testing. A pharmaceutical analysis experiment for instrumental analysis laboratory. The Chemical Educator, 15,484–487.
- Mbindyo, J. K. N., & Brown, A. K. (2010). Investigating UV-Blocking Properties of Sunscreens on the Microscale. J. Chem. Educ., 87, 1388.
- Andersen, J. E. T. (2009). Exercise in quality assurance: A laboratory exercise. Journal of Chemical Education, 86,733–737.
- Reid, K. R., Meyerhoff, M. E., & Mitchell-Koch, J. T. (2008). Salicylate detection by complexation with iron(III) and optical absorbance spectroscopy. An undergraduate quantitative analysis experiment. Journal of Chemical Education, 85, 1658–1659.
- Dwyer, T. J., Fillo, & J. D. (2006). Assaying α-dicarbonyl compounds in wine: A complementary GC-MS, HPLC and visible spectrophotometric analysis. Journal of Chemical Education, 83, 273–276.
- Mouer, H. P., Zanella, A., & Poon, T. (2006). An introduction to UV-Vis spectroscopy using sunscreens. Journal of Chemical Education, 83, 769.
- Beckers, J. L. (2004). The determination of caffeine in coffee: Sense or nonsense? Journal of Chemical Education, 81, 90–93.
- Patterson, B. M., Danielson, N. D., Lorigan, G. A., & Sommer, A. S. (2003). Analytical spectroscopy using modulator systems. Journal of Chemical Education, 80, 1460–1463.
- Byrd, H., & O’Donell, S. E. (2003). A General Chemistry Laboratory Theme: Spectroscopic Analysis of Aspirin. J. Chem. Educ., 80, 174–176.
- Aberásturi, F., Jiménez, A. I., Jiménez, F., & Arias, J. J. (2001). UV-Visible first-derivative spectrophotometry applied to an analysis of a vitamin mixture. Journal of Chemical Education, 78, 793–795.
- Aroti, A., & Leontidis, E. (2001). Simultaneous determination of the ionization constant and the solubility of sparingly soluble drug substances. A physical chemistry experiment. Journal of Chemical Education, 78, 786–788.
- Ribone, M. É., Pagani, A. P., Olivieri, A. C., & Goicoechea, H. C. (2000). Determination of the active principle in a syrup by spectrophotometry and principal component regression analysis. An advanced undergraduate experiment involving Chemometrics. Journal of Chemical Education, 77,1330– 1333.
- Aurian-Blajeni, B., Sam, J., & Sisak, M. (1999). Sweet chemistry. Journal of Chemical Education, 76, 91–92.
- McDevitt, V. L., Rodriguez, A., & Williams, K. R. (1998). Analysis of soft drinks: UV spectrophotometry, liquid chromatography and capillary electrophoresis.Journal of Chemical Education, 75, 625–628.
- Adams, P. E. (1995). Determining iron content in foods by spectrophotometry. Journal of Chemical Education, 72,649–651.
- Yarnelle, M. K., & West, K. J. (1989). Modification of an ultraviolet spectrophotometric determination of the active ingredients in APC tablets. Journal of Chemical Education, 66, 601–602.
- Stolzberg, R. J. (1986). Evaluation of derivative ultraviolet spectrometry for determining saccharin in cola and other matrices: An instrumental methods experiment. Journal of Chemical Education, 63, 351–353.
- Van Atta, R. E. (1979). Ultraviolet spectrophotometric determination of caffeine in cola Drinks: An organic analytical experiment. Journal of Chemical Education, 56, 666.
- Fernandez, L. T., Mannett, K., & Frederick H. (1978). Experiment for instrumental analysis. The determination of aspirin by ultraviolet absorption.Journal of Chemical Education, 55, 266.
- European Pharmacopoeia. (2010). European directorate for the quality of medicines and health care (7th edition), Council of Europe, Strasbourg.
- Pharmacopeia–National Formulary (USP35 – NF30). (2012). United States pharmacopeial convention, Rockville, USA.
- Under revision, 95-105%, USP Medicines Compendium (USP-MC). available at https://mc.usp.org (last accessed 06 June 2013).
- International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use. (1994). ICH guideline Q2(R1): Validation of analytical procedures: text and methodology, Geneva, Switzerland.
- Currie, L. A. (1999). Nomenclature in evaluation of analytical methods including detection and quantification capabilities. Analytica Chimica Acta, 391(2), 105-126.
